



 ZNG Medical
ZNG Medical










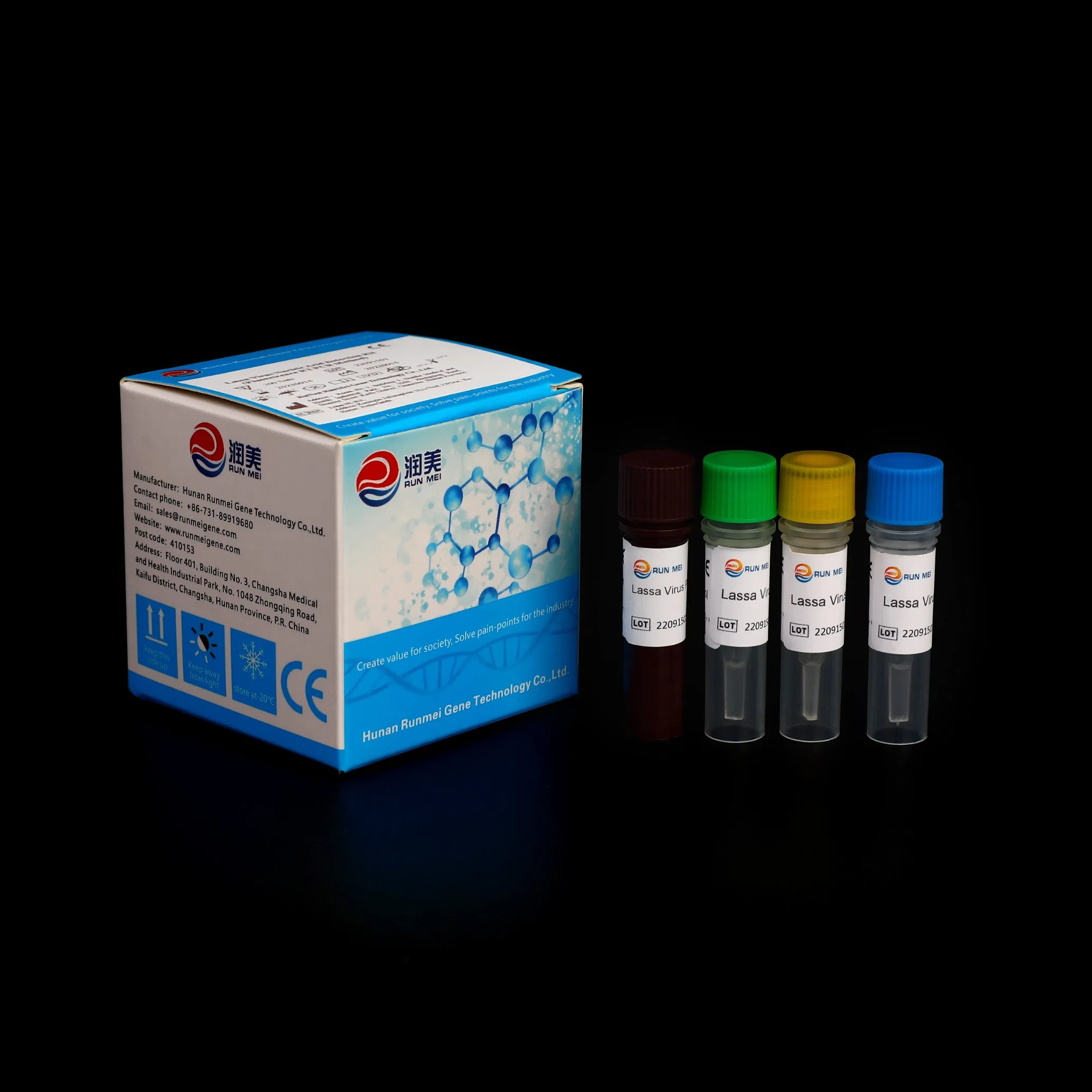
Lassa Virus Nucleic Acid Detection Kit (Fluorescence RT-PCR Method)
This kit is used for the detection of Lassa Virus Nucleic Acid and assisted diagnosis and epidemiological surveillance of Lassa Virus. For professional use only.
| Composition | 20 Tests/Kit | 50 Tests/Kit | 200 Tests/Kit |
|---|---|---|---|
| Lassa Virus PCR Reaction Solution | 300 μL*1 tube | 750 μL*1 tube | 750 μL*4 tube |
| Lassa Virus Enzyme Mixture | 100 μL*1 tube | 250 μL*1 tube | 250 μL*4 tube |
| Lassa Virus Negative Control | 100 μL*1 tube | 100 μL*1 tube | 100 μL*4 tube |
| Lassa Virus Positive Control | 100 μL*1 tube | 100 μL*1 tube | 100 μL*4 tube |
| Instructions | 1 | 1 | 1 |
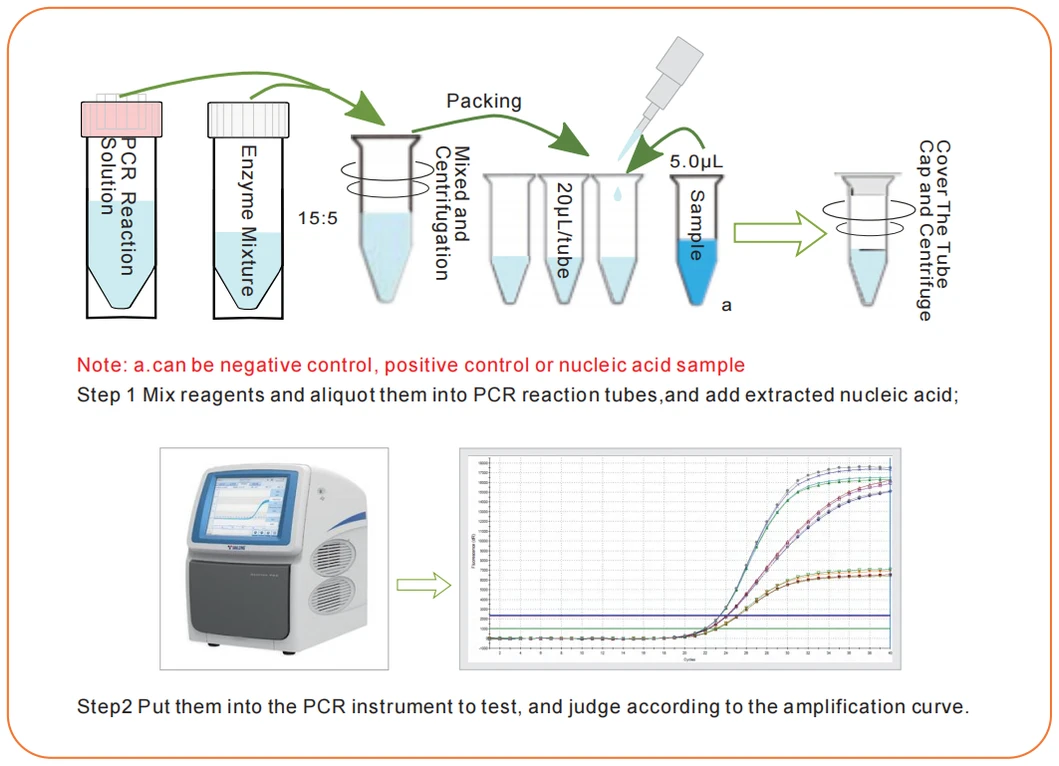
Storage Conditions and Expiring Date:
Applicable Instrument:
Suitable for ABI series, Bio-Rad series, Agilent Stratagene MX series, Roche LightCycler R480, Cepheid SmartCycler, Rotor-Gene series and other multi-channel real-time quantitative PCR instruments.


Founded in 2018, the company is based in the fields of molecular biology and diagnostics, and has a unique "product + service" model. Partners include well-known institutions and universities worldwide.
The company has a R&D team consisting of experts from worldwide, and has established a high-standard 100,000-grade GMP production workshop and obtained the ISO9001 quality management system certification. Based on the business philosophy of "trust comes from quality", we focus on global needs and are dedicated to providing customers with quality products and good technical services.



