

 ZNG Medical
ZNG Medical











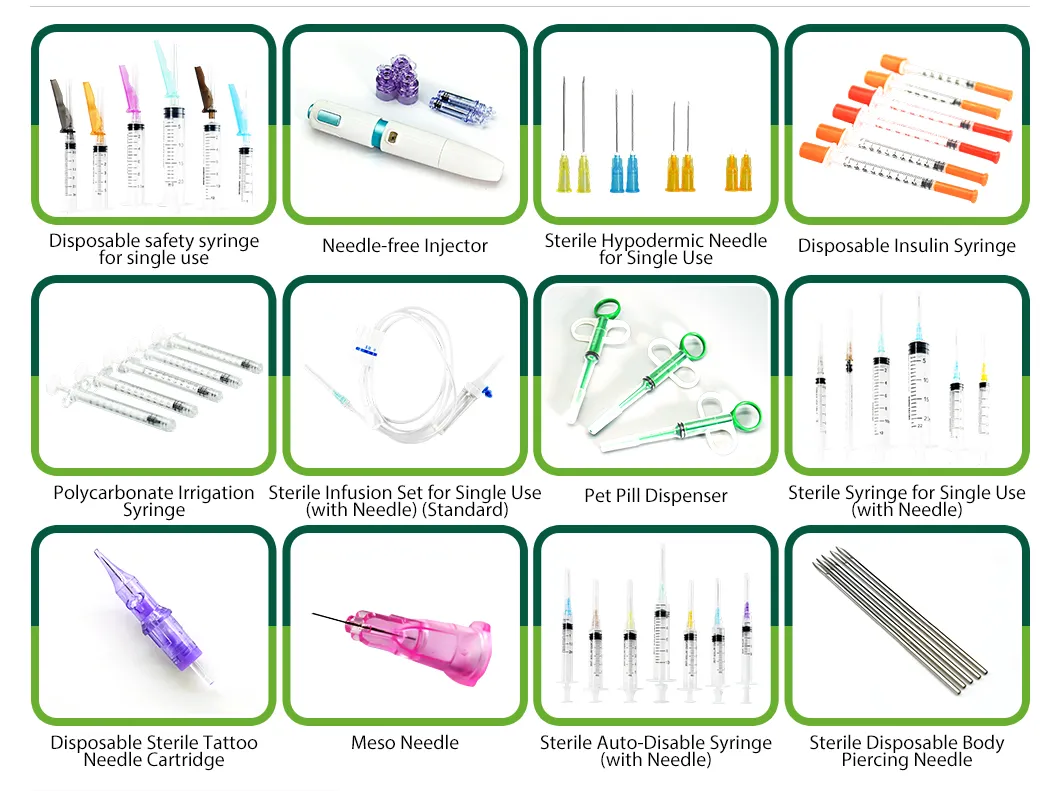

Established in 2005, we are a manufacturer specialized in disposable sterile medical consumables and related single-use devices. We focus on consistent quality, scalable production, and reliable delivery for global OEM/ODM customers. Our site covers approximately 32,000 m2 with a built-up area of over 60,000 m2.
We operate more than 12,000 m2 of ISO Class 8 cleanroom facilities. Sterilization is supported by 8 EO sterilization cabinets. For quality assurance, we run an in-house testing center capable of biological, chemical, and physical testing, with 160+ sets of production equipment and 70+ sets of testing/QC equipment.
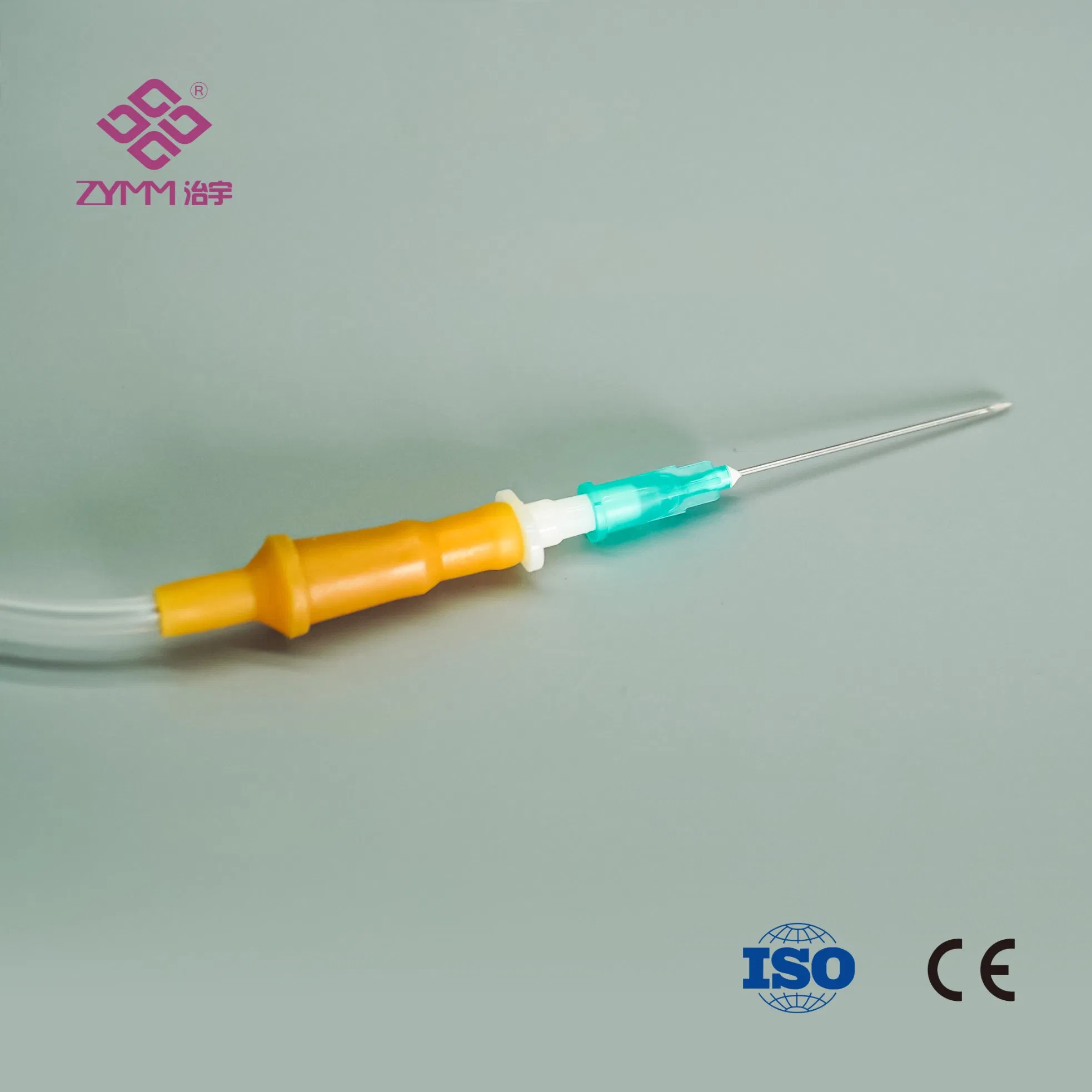
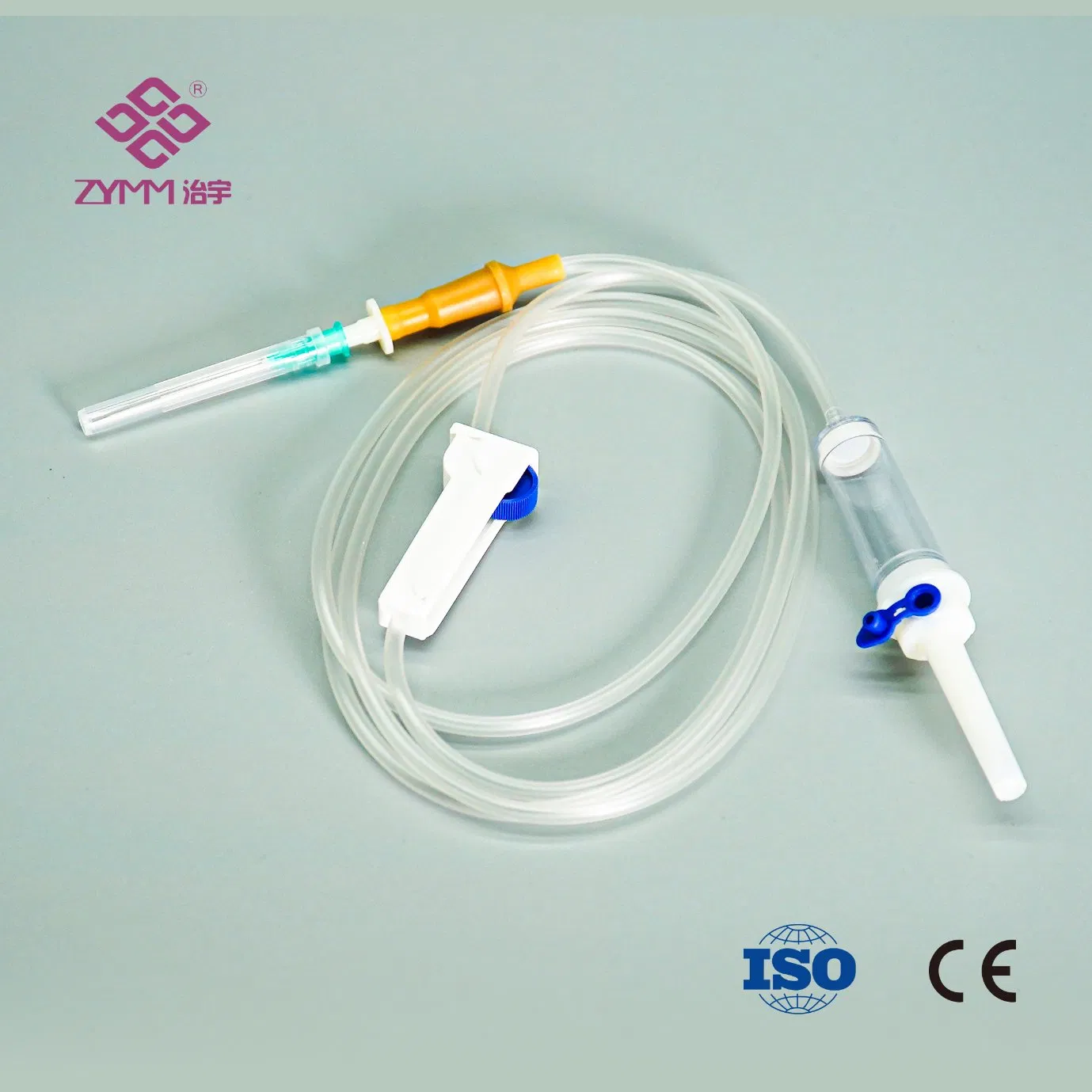
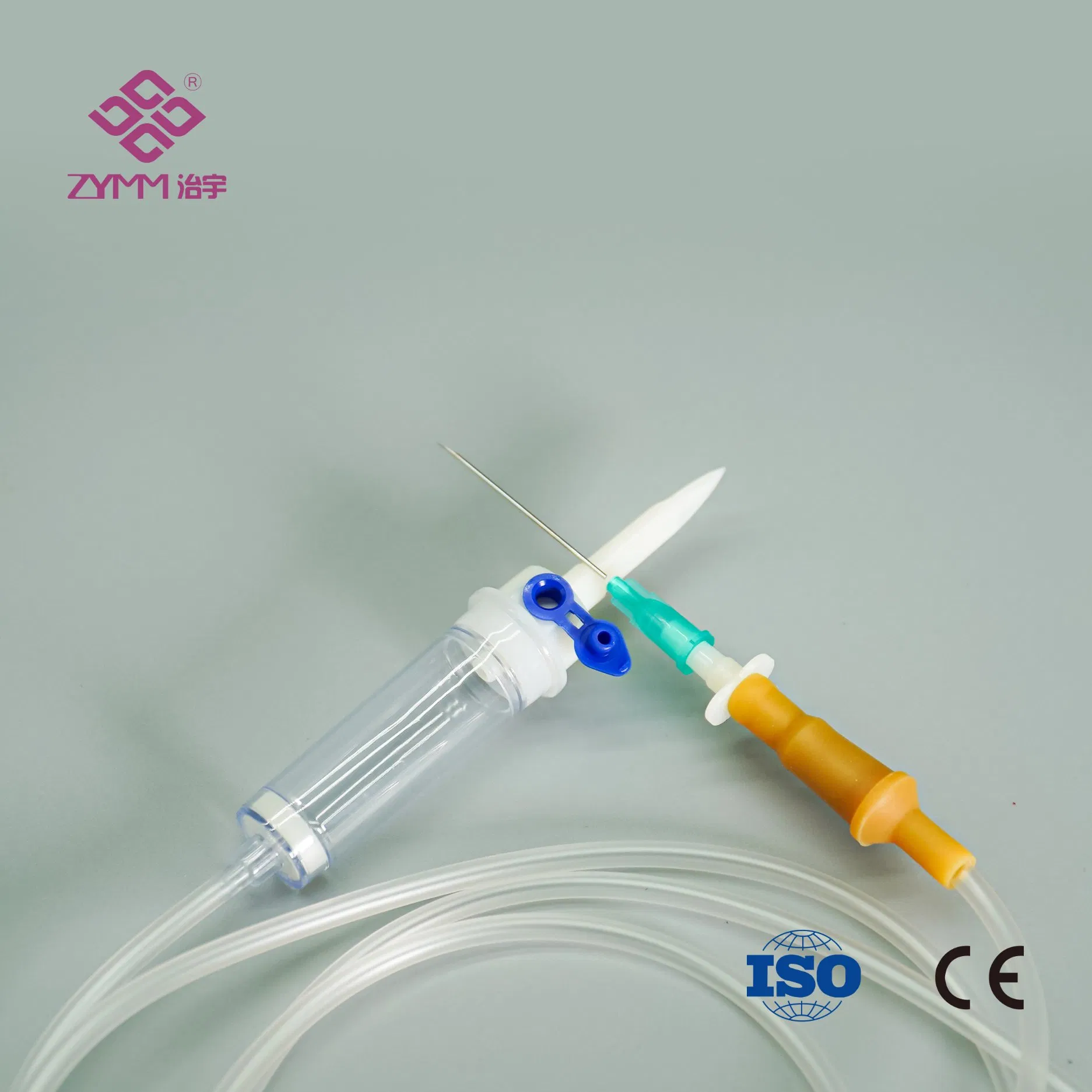
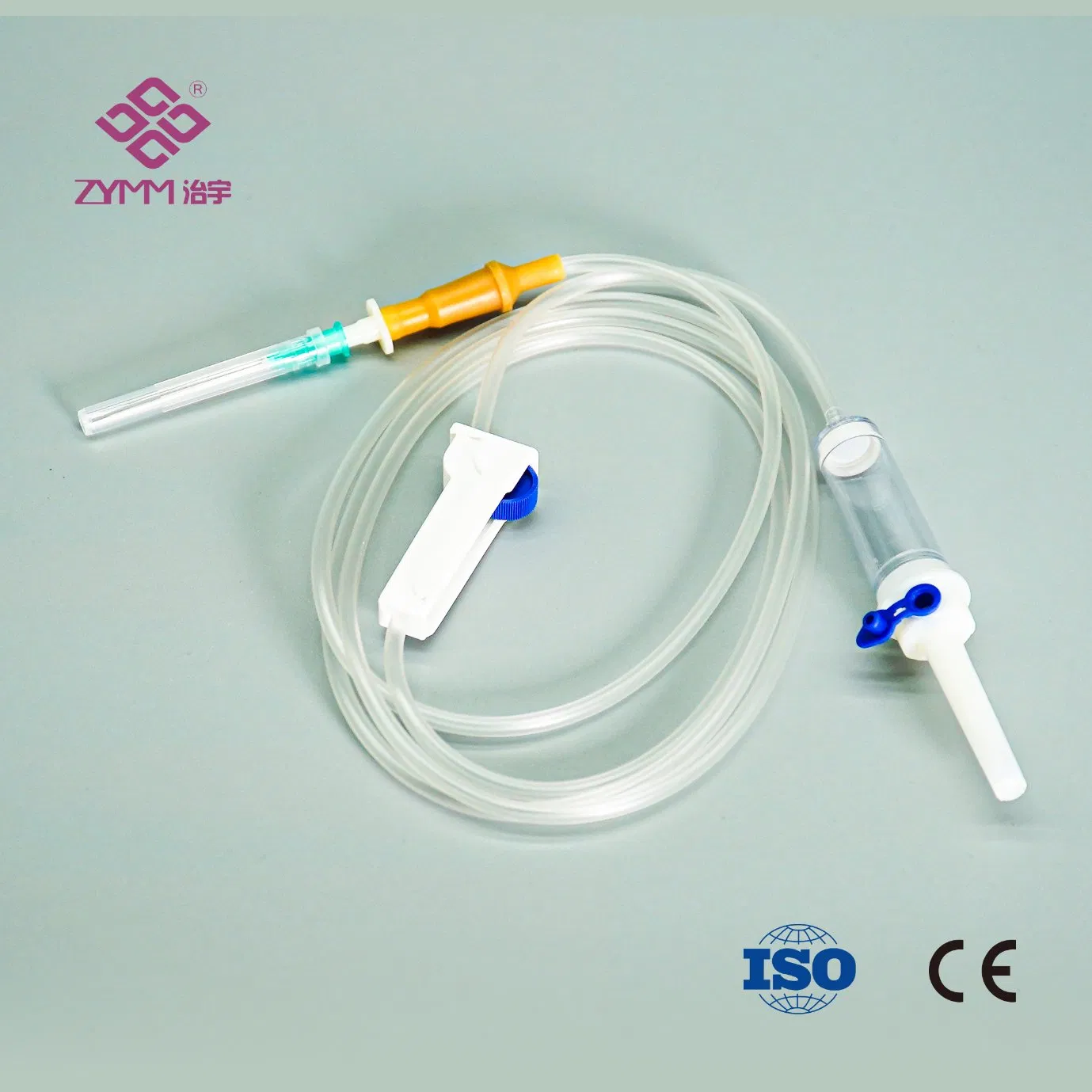
Our annual output of syringes exceeds 1 billion pieces, infusion sets reach around 100 million pieces annually, and hypodermic needles can reach up to 2 million pieces per day. We support multi-SKU, multi-batch purchasing plans with reliable lead times.



We operate an ISO 13485 certified quality management system, hold EU MDR (EU) 2017/745 QMS certificates (TUV Rheinland), maintain FDA establishment registration, and have 510(k) clearance for specific product families. OEM/ODM cooperation includes customized configurations, private labeling, artwork support, and multi-language packaging.

