 ZNG Medical
ZNG Medical
 ZNG Medical
ZNG Medical
Precision-engineered products for clinical pathology and microbiological applications.

Yas Rgl 4f 5f 6f Contrast Media Delivery Pathway Radiology Angiographic Catheter

Angiography Catheter Vertebral Vascular System During Diagnostic Endovascular Procedures
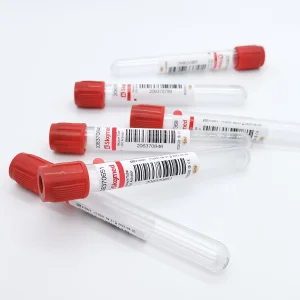
Clot Activator Vacuum Blood Collection Tube 5ml Factory Supply Directly
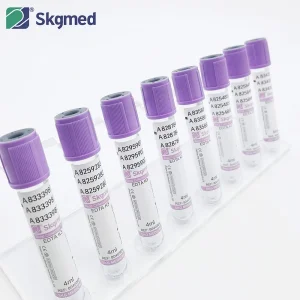
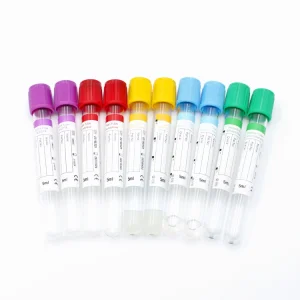
Disposable Medical Test Pet Glass Plain/Procoagulation/Gel & Clot Activator
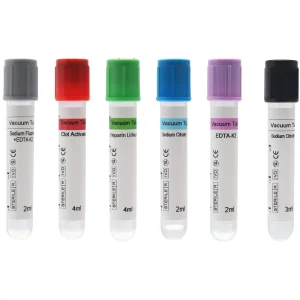
Siny Disposable All Type Micro EDTA Yellow Clot Activator Vacuum Blood Collection Tube
EDTA Micro Blood Collection Tube for Children & Geriatric Patient

Separate Gel and Clot Activator Blood Collection Tube with CE Approval
Understanding the vital role of microbiological growth environments in modern industry.
The global market for bacteriological culture media is witnessing an unprecedented surge, driven by the escalating demand for rapid diagnostic testing and the expansion of the biopharmaceutical industry. As a cornerstone of clinical microbiology, culture media serve as the fundamental substrate for isolating, identifying, and testing the antibiotic susceptibility of pathogenic microorganisms. From hospital laboratories to food safety auditing firms, the requirement for high-purity, standardized media has never been more critical.
In the current industrial climate, the transition from traditional Dehydrated Culture Media (DCM) to Ready-to-Use (RTU) formats is a significant trend. Global laboratories are increasingly prioritizing workflow efficiency and the reduction of human error. As a leading manufacturer in China, we integrate advanced biotechnology with large-scale manufacturing to provide both standard formulations and customized media that meet the rigorous standards of the European Pharmacopoeia (EP) and the United States Pharmacopeia (USP).
The global microbiology culture market is projected to reach billions by 2030. The shift toward automated inoculation systems is driving the need for extremely consistent media batches.
With the rise of specialized cell therapies, bacteriological media are now being adapted for quality control in cleanroom environments, ensuring zero contamination in life-saving drugs.
Emerging markets in Southeast Asia and Africa are focusing on epidemiological surveillance, creating massive demand for affordable, high-quality export-grade media.
Leveraging supply chain integration and technological innovation.
China's medical device sector has evolved from simple assembly to high-end R&D. Our facilities utilize AI-driven quality control to monitor production parameters in real-time, ensuring every petri dish or culture tube meets international benchmarks.
By optimizing the raw material sourcing for agar, peptones, and yeast extracts, Chinese manufacturers offer a 30-50% cost advantage without compromising the biological performance of the media.
With specialized cold-chain logistics and vacuum-sealed packaging, we ensure that the shelf-life and stability of our bacteriological media are preserved from our factory in Zhejiang to your laboratory anywhere in the world.
A legacy of excellence in medical device manufacturing since 1996.
Zhejiang ZNG Medical, founded in 1996, stands as a leading force in the medical device industry. We are dedicated to the research, development, and production of high-precision injection and diagnostic products. With a foundation built on ISO 9001 quality systems and CE product certification, we provide the global market with secure, efficient, and reliable medical solutions.
Our expansive product portfolio includes specialized Needles: Pen needles, blood collection needles, spinal and dental needles, epidural, chiba, and biopsy needles. We also specialize in the development of custom needles according to specific customer drawings and mold-making requirements.
Our Mission: At ZNG Medical, we are committed to public health. By actively listening to our global partners, we continuously improve our existing lines and innovate new products to meet the evolving needs of modern medicine.
Transparency and efficiency from the first inquiry to final delivery.
High-tech production lines ensuring sterile and precise medical instruments.
Producing Cannula
Injection Molding
Assembling

Sterilized by EO
Stay ahead with insights into localized application scenarios and procurement strategies.
Localized Application Scenarios: In the European market, there is a sharp increase in the use of specialized chromogenic media for the rapid detection of MRSA and other multi-drug resistant organisms. Conversely, in the Latin American and Southeast Asian agricultural sectors, bacteriological culture media are being utilized heavily for water quality testing and soil microbiome analysis to enhance crop yields. As an exporter, we provide localized formulations that cater to the specific microbial strains prevalent in different geographical regions.
Global Procurement Strategy: Savvy procurement officers are moving away from single-source dependencies. By partnering with a large-scale manufacturer like ZNG Medical in China, global firms gain access to a redundant supply chain that can handle massive volume spikes during public health crises. Our commitment to E-E-A-T principles means every certificate of analysis (COA) is backed by rigorous internal testing and external audits.
The convergence of Artificial Intelligence and microbiology is the next frontier. We are currently exploring the development of media optimized for digital imaging systems, where AI algorithms can identify colony morphology faster than the human eye. This technological edge makes our products not just consumables, but essential components of the 21st-century diagnostic ecosystem.
Explore our high-performance catheters and surgical diagnostic tools.