1 / 5
 ZNG Medical
ZNG Medical










The micro catheter is intended for the infusion of contrast media, medicine, or embolic material into coronary vasculatures. It is also designed to provide support for the placement of guide wires or guide wire exchange.

Consists of a tube hub, protect sheath, catheter body, and developing ring. The materials include Pebax, Eurelon, 304V Stainless steel, PTFE coating, Platinoiridium, TPU, PC, and Hydrophilic coating. Packaged in a paper-plastic pouch and EO sterilized.
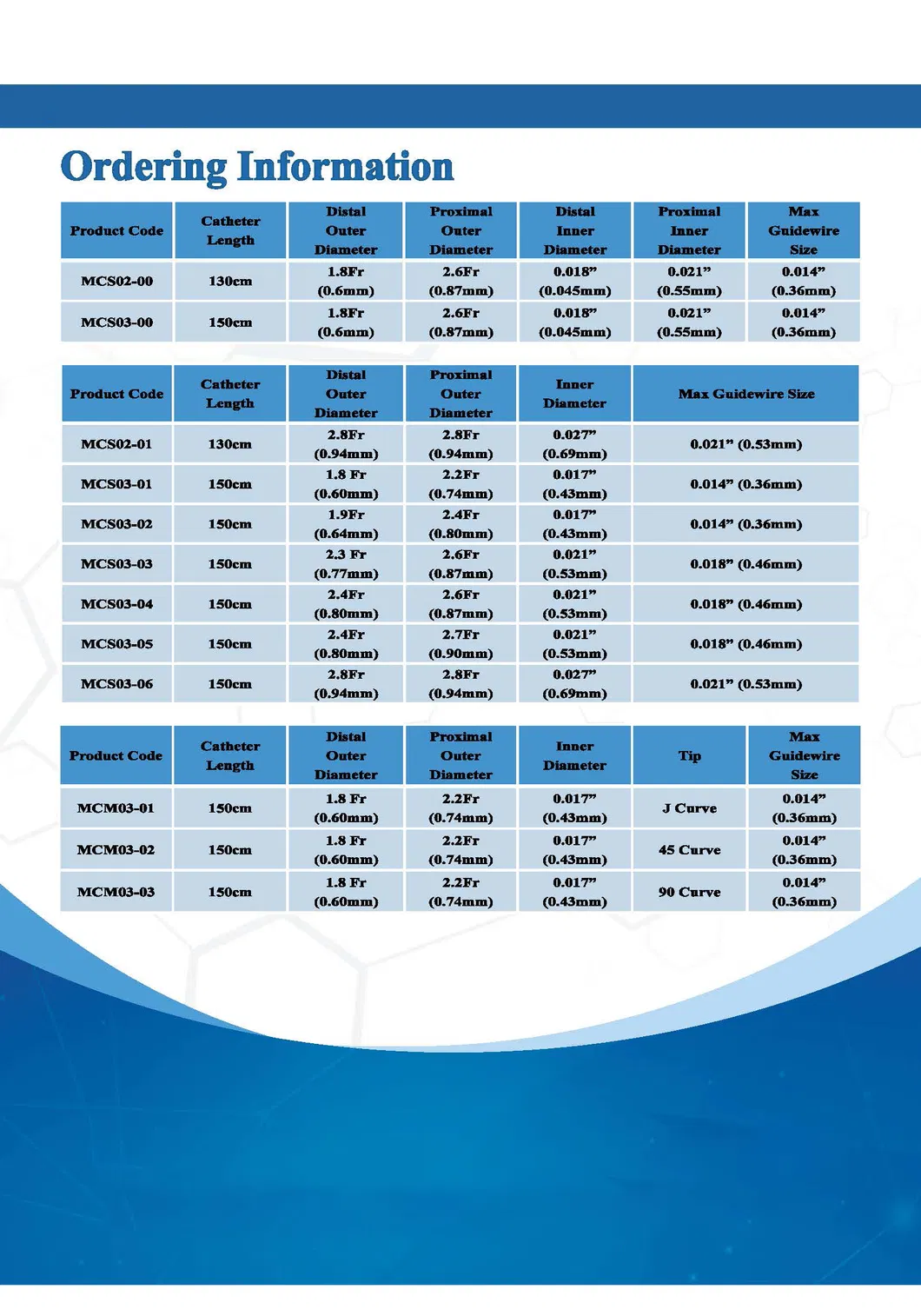
| REF NO. | Outer Diameter | Inner Diameter | Usable Length | Rec. GW | ||
|---|---|---|---|---|---|---|
| Distal Shaft | Proximal Shaft | Tip | Shaft | |||
| MCS02-6 | 0.93mm (2.8Fr) | 1.00mm (3.0Fr) | 0.68mm (0.027") | 0.68mm (0.027") | 130cm | 0.53mm (0.021") |
| MCS03-6 | 0.93mm (2.8Fr) | 1.00mm (3.0Fr) | 0.68mm (0.027") | 0.68mm (0.027") | 150cm | 0.53mm (0.021") |